The project delivered a validated tool compound and a macrocyclic lead with improved potency and genuine TRPML1 selectivity. BioAscent’s scientific expertise and screening deck capability, enabled the identification of novel, truly selective agonists in a target space where published reference compounds show significant non-specific activity. This provided the client with robust, differentiated starting points and avoided the risk of pursuing misleading or non‑selective chemical matter.
The transient receptor potential channel mucolipin 1 (TRPML1) ion channel plays an important role in neurological development and function and is the most characterised member of the mucolipin subfamily of transient receptor potential cation channels (TRPML) localised to lysosomes. Modulating TRPML1 function is of increasing interest in neurodegeneration and lysosomal biology, but the intracellular localisation and reported non-specificity of published agonists present challenges for assay development and novel hit finding.
The client, a US-based biotech working in the neurodegeneration space, required rapid progression of a TRPML1 agonist programme to support target validation and early drug discovery.
In early experiments commercially available TRPML1 activators ML‑SA5 and ML‑SA11 displayed similar potency in both overexpressing (OE) and knock‑out (KO) HEK293_TRPML1 cell lines. This non‑specific activity made it difficult to identify true TRPML1‑mediated Ca²⁺ release and threatened to compromise screening fidelity.
TRPML1 assay development
BioAscent scientists developed, optimised, and validated real-time Ca²⁺ flux assays on HEK293_TRPML1 OE and KO cell lines using FLIPR Penta assay technology. The assays were validated using two disclosed TRPML1 activators, ML-SA5 and ML-SA1 (n = 4). To ensure TRPML1-dependent signalling could be confidently resolved, extracellular Ca²+ free conditions were developed to maximise assay specificity (Figure 1).
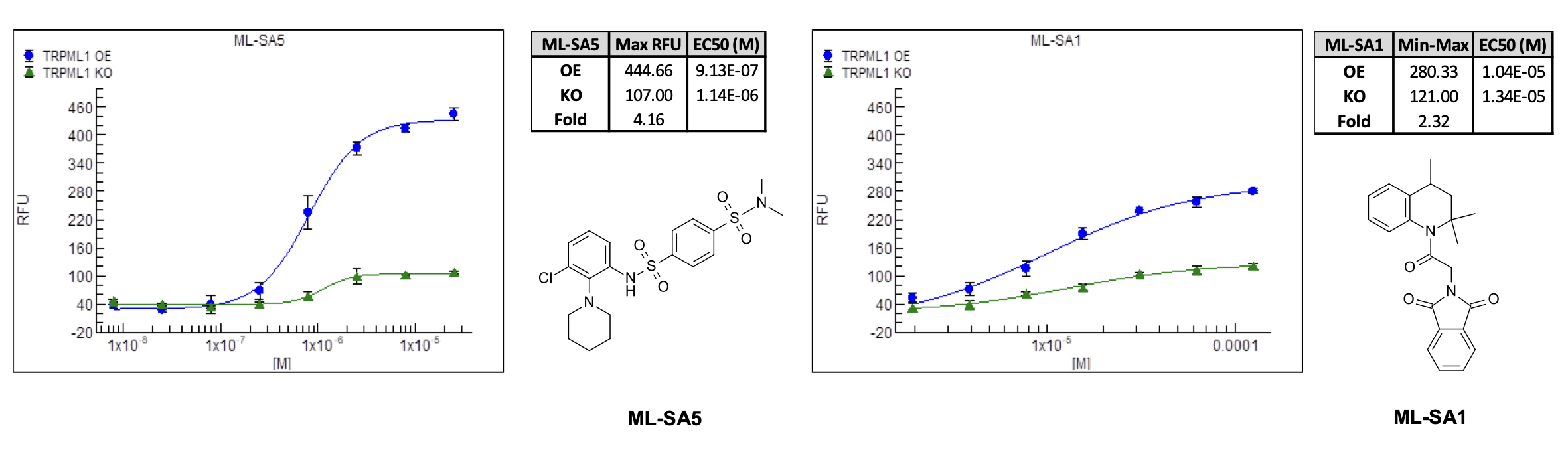
Figure 1. The TRPML1 activators ML-SA5 and ML-SA1 cause a dose-dependent increase in cytosolic Ca2+ in TRMPL1 OE cells with minimal effect upon the TRMPL1 KO cell line after removal of extracellular Ca2+.
High‑throughput screening of >100,000 compounds
The BioAscent Library of >100,000 compounds was screened against the TRPML1 OE cell line at a single concentration of 10 µM (Figure 2).
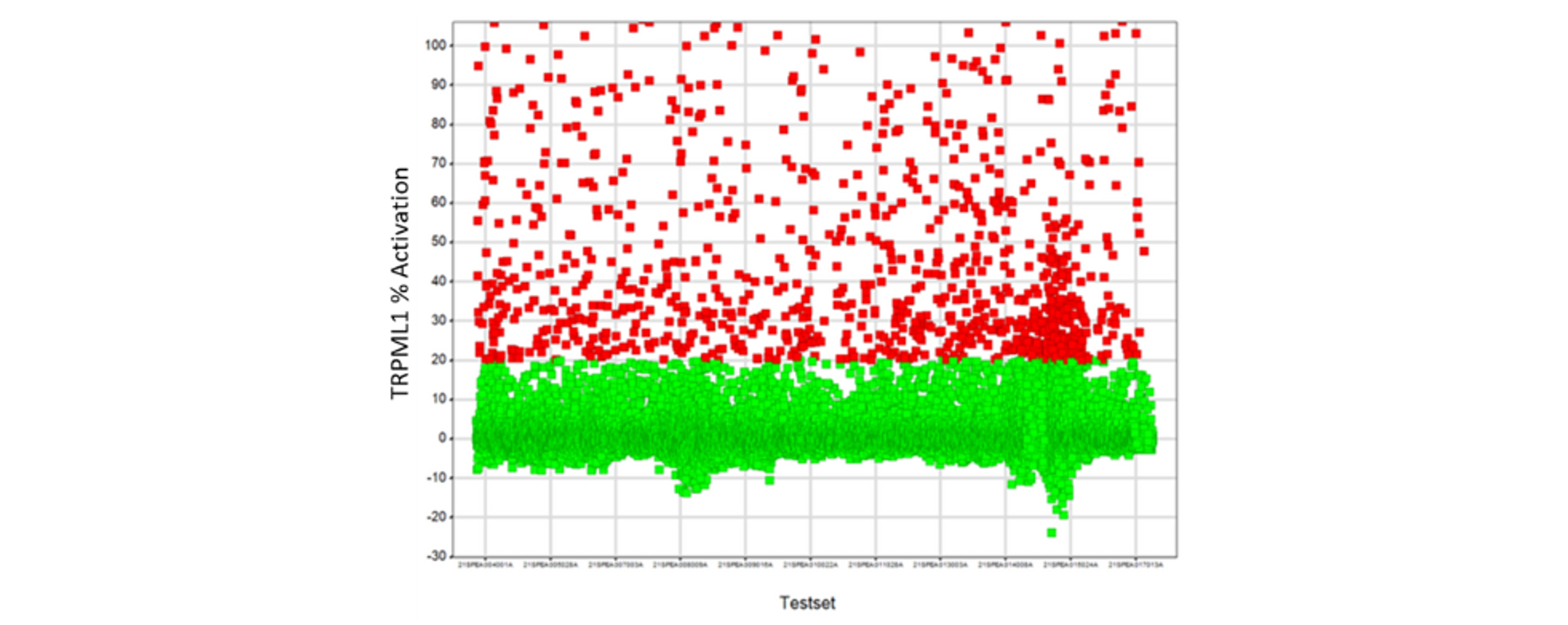
Figure 2: TRPML1 OE primary screen. ~1700 compound eliciting a response of ≥15% activation relative to 25 µM ML-SA5 control are highlighted in red and were progressed to active confirmation/deselection.
Hit confirmation and triage
109 compounds were tested in 7-point and then 11-point dose–response curves in both TRPML1 OE and KO cells.
Two compounds, BCC0092140 and BCC0044046, demonstrated:
High potency in TRPML1‑OE cells
Minimal/no activity in TRPML1‑KO cells
Nanomolar to low micromolar EC₅₀ values
These hits were taken forward for characterisation and further optimisation.
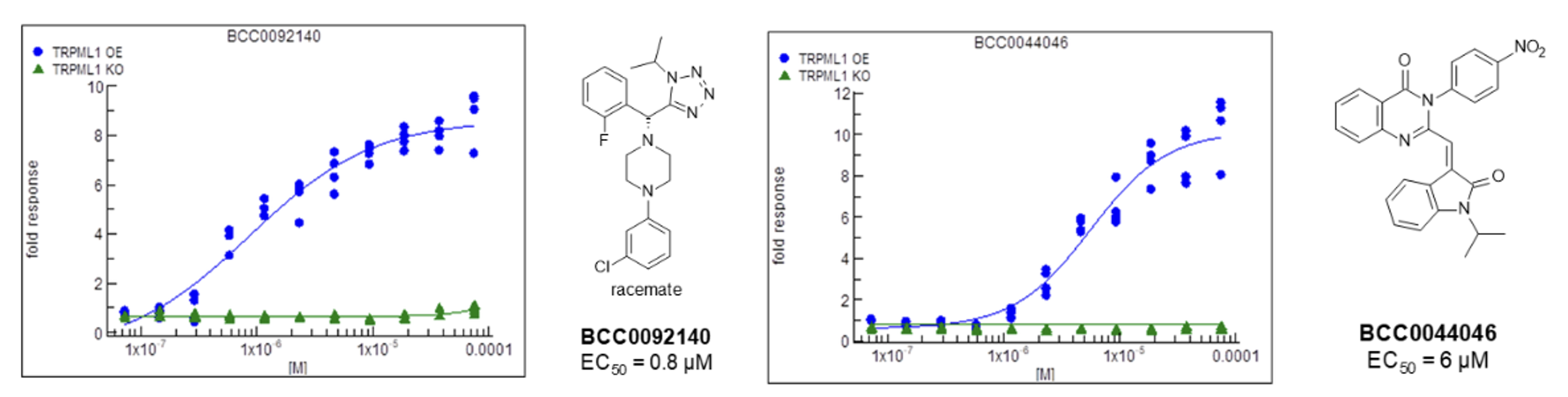
Figure 3: Dose-response data for the two most promising compounds.
Chiral separation and ADME profiling
After resynthesis and chiral separation of BCC0092140:
Enantiomer 1a showed an EC₅₀ of 0.481 µM in TRPML1‑OE cells
Selectivity >100‑fold relative to KO cells
In vitro ADME data were collected for both enantiomers, showing viable early stage developability (Table 1):
Encouragingly, the compounds demonstrated high permeability and were not efflux substrates in a MDCK-MDR1 over-expressing cellular assay
Both compounds had high plasma protein binding across species and high brain tissue binding in mice
Stability in hepatocytes varied across species. Half-life and clearance were moderate in human but poor in rat, with scope for improvement providing a focus for further development
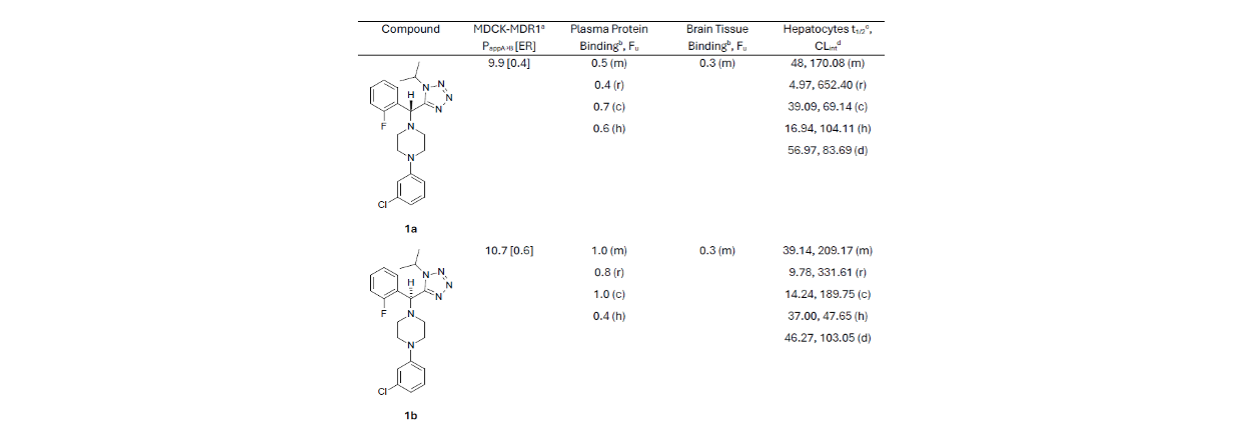
Table 1. In vitro ADME data for compounds 1a and 1b. a (*10-6 cm/s); b %; m: mouse, r: rat, d: dog; c: cynomolgus monkey, h: human; c minutes; d μL/min/kg.
Structure‑based design of macrocyclic compounds
Computational docking studies confirmed the absolute configuration of the active enantiomer 1a, showing a good fit in the binding pocket occupied by ML-SA1 rather than at the orthosteric binding site occupied by the endogenous agonist (Figure 4). The fluorophenyl and the isopropyl tetrazole moieties of compound 1a retained some degree of free rotation in our model. Structure-based design was used to guide macrocycle design to reduce the number of available conformations and unfavourable entropic contributions to the binding energy with the hypothesis that this may improve potency.
Three macrocycles were synthesised which retained potency, with compound 18 showing improved potency (EC₅₀ of 0.270 µM) in the TRPML1 OE cell line, with no activity in the TRPML1 KO cells (Table 2).
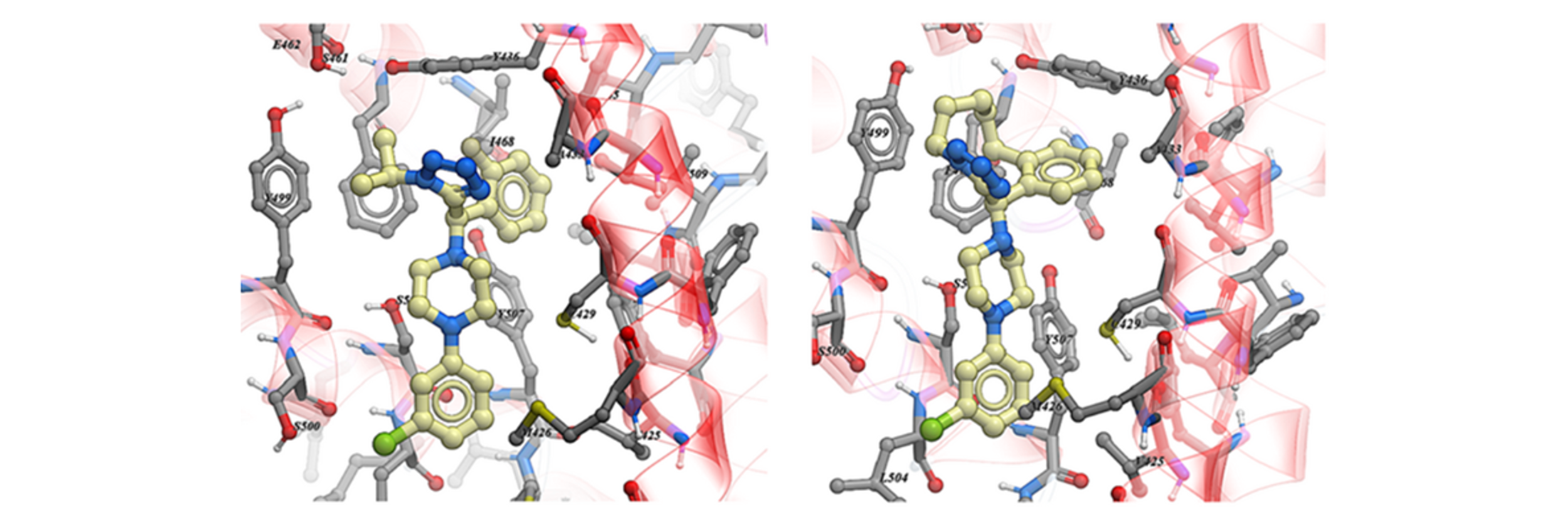
Figure 4. Docking pose of the most active BCC0092140 enantiomer 1a (left panel) and (R)-Compound 19 (right panel) into the TRPML1 Cryo-EM structure.
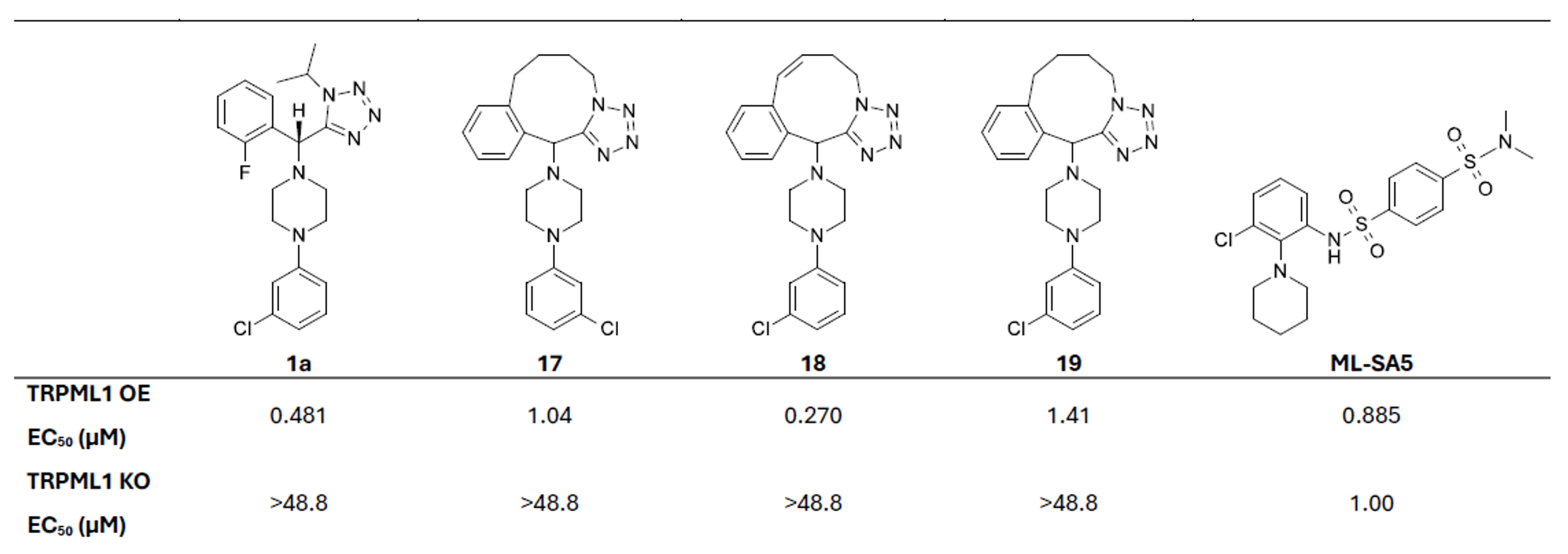
Table 2. EC50 data for synthesised macrocycles in TRPML1 OE and KO cell lines.
We enabled the identification and optimisation of novel TRPML1 agonists by applying BioAscent’s expertise in assay development, HTS and in silico discovery, as well as our diversity screening collection. This effort delivered compound 18, a promising, genuinely TRPML1-selective lead suitable for further optimisation.
By resolving the nonspecific activity observed with published TRPML1 agonists and filtering out the large proportion of misleading hits, the project provided the client with high-quality, differentiated starting points. This substantially reduced the design effort that would otherwise have been required to engineer selectivity from poor-quality chemical matter and helped de-risk the programme at an early stage.
Learn more about BioAscent’s integrated drug discovery expertise here.
Spix, B., Butz, E.S., Chen, CC. et al. Nat. Commun. 2022, 13, 318. https://doi.org/10.1038/s41467-021-27860-x
Discovery and characterization of novel TRPML1 agonists. Peng, X. et al. Bioorg. Med. Chem. Lett. 2024, 98, 129595. https://doi.org/10.1016/j.bmcl.2023.129595




